|
|
|
|
Year of first isolation: |
2012 |
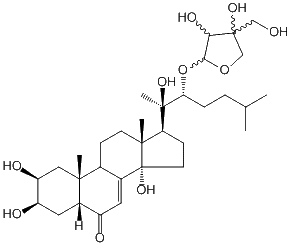
| Formula: | C32H52O10 |
| Molecular weight: | 596 |
| Occurence in plants: |
Serratula wolffii [Asteraceae] »  ![Wikipedia: Serratula wolffii [Asteraceae]](/images/wikipedia.png)
|
| Occurence in animals: |
|
|
|
| |
| Canonical SMILES | | Isomeric SMILES
[ PubChem: search | XML ]
[ ChemSpider: search ] | C1[C@]2([C@@H](C[C@H]([C@H]1O)O)C(=O)C=C1[C@@H]2CC[C@]2([C@]1(CC[C@@H]2[C@](C)([C@@H](CCC(C)C)OC1C(C(CO1)(O)CO)O)O)O)C)C » 
| | IUPAC Name | (2S,3R,5R,9R,10R,13R,14S,17S)-17-((2R,3R)-3-((3,4-dihydroxy-4-(hydroxymethyl)tetrahydrofuran-2-yl)oxy)-2-hydroxy-6-methylheptan-2-yl)-2,3,14-trihydroxy-10,13-dimethyl-1,2,3,4,5,9,10,11,12,13,14,15,16,17-tetradecahydro-6H-cyclopenta[a]phenanthren-6-one | | CAS-RN | | | PubChem CID | | InChiKey
[ ChemIDPlus: search ] | IZZSIVXWZMRKEQ-MYTIXXAXSA-N | | InChI | InChI=1S/C32H52O10/c1-17(2)6-7-25(42-27-26(37)31(39,15-33)16-41-27)30(5,38)24-9-11-32(40)19-12-21(34)20-13-22(35)23(36)14-28(20,3)18(19)8-10-29(24,32)4/h12,17-18,20,22-27,33,35-40H,6-11,13-16H2,1-5H3/t18-,20-,22+,23-,24-,25+,26?,27?,28+,29+,30+,31?,32+/m0/s1 |
| |
| HR-ESI-MS | 597.36234, calculated 597.36332 for C32H53O10 | | CI-MS (NH3) m/z | | | ESI-MS m/z (relative intensity %) | 597 (0.5) (M+H)+, 465 (100) (M+H-C5H8O4), 447 (28) (M+H-C5H8O4-H2O), 429 (15) (M+H-C5H8O4-2H2O), 299 (5) (C19H23O3) |
|
|
| CD3OD | | 01 | 37.5 | | 02 | 68.9 | | 03 | 68.7 | | 04 | 33.0 | | 05 | 52.0 | | 06 | 206.6 | | 07 | 122.3 | | 08 | 168.1 | | 09 | 35.2 | | 10 | 39.4 | | 11 | 21.7 | | 12 | 32.8 | | 13 | 48.8 | | 14 | 85.4 | | 15 | 31.9 | | 16 | 21.5 | | 17 | 51.2 | | 18 | 18.3 | | 19 | 24.5 | | 20 | 77.3 | | 21 | 22.6 | | 22 | 90.2 | | 23 | 30.8 | | 24 | 36.9 | | 25 | 29.4 | | 26 | 22.8 | | 27 | 23.5 | | Api-1' | 113.0 | | Api-2' | 77.9 | | Api-3' | 80.3 | | Api-4' | 74.7 | | Api-5' | 64.8 |
|
| CD3OD | | 01-Ha | 1.43 (dd, 13.1, 12.0) | | 01-He | 1.79 (m) | | 02-Ha | 3.84 (ddd, 12.0, 4.1, 3.4) | | 03-He | 3.95 (m) | | 04-Ha | 1.75 (m) | | 04-He | 1.70 (m) | | 05-H | 2.38 (dd, 13.0, 4.1) | | 07-H | 5.81 (d, 2.6) | | 09-H | 3.15 (ddd, 10.8, 7.4, 2.5) | | 11-Ha | 1.69 (m) | | 11-He | 1.81 (m) | | 12-Ha | 2.11 (td, 12.9, 4.7) | | 12-He | 1.88 (ddd, 12.9, 4.7, 1.8) | | 15-Ha | 1.58 (m) | | 15-Hb | 1.98 (m) | | 16-Ha | 1.67 (m) | | 16-Hb | 2.01 (m) | | 17-H | 2.30 (t, 9.0) | | 18-Me | 0.88 (s) | | 19-Me | 0.97 (s) | | 21-Me | 1.195 (s) | | 22-H | 3.35 (dd, 10.6, 1.6) | | 23-Ha | 1.37 (m) | | 23-Hb | 1.61 (m) | | 24-Ha | 1.25 (m) | | 24-Hb | 1.51 (m) | | 25-H | 1.55 (qui, 6.7) | | 26-Me | 0.92 (d, 6.7) | | 27-Me | 0.93 (d, 6.7) | | [Api] H-1' | 4.98 (d, 3.6) | | [Api] H-2' | 3.96 (d, 3.6) | | [Api] H-4' | 3.79 (d, 9.6) | | [Api] H-4'' | 4.10 (d, 9.6) | | [Api] H-5' | 3.55 (d, 11.5) | | [Api] H-5'' | 3.58 (d, 11.5) |
|
| |
| M.P. | °C ; | | [α]D27 | +9 ° (c 0.1 ; MeOH) | | IR (KBr) ν max (cm-1) | | | UV (MeOH) λ max (log ε) | 242 (2.81) ; |
| |
| |
| |
| |
Permanent link to this datasheet: PONASTERONE A 22-APIOSIDE
|
|




![Wikipedia: Serratula wolffii [Asteraceae]](/images/wikipedia.png)

