|
|
|
|
Year of first isolation: |
1977 |
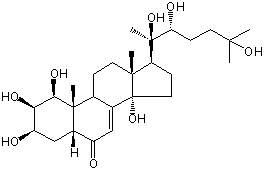
| Formula: | C27H44O8 |
| Molecular weight: | 496 |
| Occurence in plants: |
Rhaponticum integrifolium [Asteraceae] »  ![Wikipedia: Rhaponticum integrifolium [Asteraceae]](/images/wikipedia.png)
Serratula xeranthemoides [Asteraceae] »  ![Wikipedia: Serratula xeranthemoides [Asteraceae]](/images/wikipedia.png)
Silene nutans [Caryophyllaceae] »  ![Wikipedia: Silene nutans [Caryophyllaceae]](/images/wikipedia.png)
Silene otites [Caryophyllaceae] »  ![Wikipedia: Silene otites [Caryophyllaceae]](/images/wikipedia.png)
Lychnis flos-culculi [Caryophyllaceae] »  ![Wikipedia: Lychnis flos-culculi [Caryophyllaceae]](/images/wikipedia.png)
Silene viridiflora [Caryophyllaceae] »  ![Wikipedia: Silene viridiflora [Caryophyllaceae]](/images/wikipedia.png)
Leuzea carthamoides [Asteraceae] »  ![Wikipedia: Leuzea carthamoides [Asteraceae]](/images/wikipedia.png)
|
| Occurence in animals: |
|
|
|
| |
| Canonical SMILES | CC12CCC3C(=CC(=O)C4C3(C(C(C(C4)O)O)O)C)C1(CCC2C(C)(C(CCC(C)(C)O)O)O)O | Isomeric SMILES
[ PubChem: search | XML ]
[ ChemSpider: search ] | C[C@]12CC[C@H]3C(=CC(=O)[C@H]4[C@@]3([C@@H]([C@@H]([C@@H](C4)O)O)O)C)[C@@]1(CC[C@@H]2[C@](C)([C@@H](CCC(C)(C)O)O)O)O » 
[C@@H]1([C@]2([C@@H](C[C@H]([C@H]1O)O)C(=O)C=C1[C@@H]2CC[C@]2([C@]1(CC[C@@H]2[C@](C)([C@@H](CCC(C)(C)O)O)O)O)C)C)O » 
| | IUPAC Name | (1S,2R,3R,5R,9R,10R,13R,14S,17S)-1,2,3,14-tetrahydroxy-10,13-dimethyl-17-[(2R,3R)-2,3,6-trihydroxy-6-methylheptan-2-yl]-2,3,4,5,9,11,12,15,16,17-decahydro-1H-cyclopenta[a]phenanthren-6-one | | CAS-RN | | | PubChem CID | 16038795 | InChiKey
[ ChemIDPlus: search ] | VJRBXZFHKYDEQV-KPHYXMJDSA-N | | InChI | InChI=1S/C27H44O8/c1-23(2,33)9-8-20(30)26(5,34)19-7-11-27(35)15-12-17(28)16-13-18(29)21(31)22(32)25(16,4)14(15)6-10-24(19,27)3/h12,14,16,18-22,29-35H,6-11,13H2,1-5H3/t14-,16-,18+,19-,20+,21+,22+,24+,25+,26+,27+/m0/s1 |
| |
| CI-MS (NH3) m/z | 497 (MH)+, 479, 461, 443, 425, 379, 361, 345, 343 | | EI-MS m/z (relative intensity %) | 478 (14), 460(20), 442 (94), 424 (33), 409 (10), 391 (6), 379 (15), 374 (12), 368 (42), 361 (100), 343 (90), 325 (30), 316 (25), 301 (15), 283 (24), 143 (12), 125 (12), 99 (49), 81 (27), 69 (27) | | HR-MS | |
|
|
| CD3OD | | 01 | 76.43 | | 02 | 70.98 | | 03 | 68.50 | | 04 | 43.80 | | 05 | 46.79 | | 06 | 205.57 | | 07 | 122.17 | | 08 | 167.19 | | 09 | 35.65 | | 10 | | | 11 | 21.91 | | 12 | 32.54 | | 13 | ~49.0 | | 14 | 85.10 | | 15 | 31.80 | | 16 | 21.40 | | 17 | 50.58 | | 18 | 18.02 | | 19 | 20.01 | | 20 | 77.89 | | 21 | 21.05 | | 22 | 78.43 | | 23 | 27.36 | | 24 | 42.38 | | 25 | 71.29 | | 26 | 29.70 | | 27 | 28.97 |
|
| C5D5N | | 01-Ha | -- | | 01-Hb | 4.15 | | 02-Ha | 4.15 | | 03-He | 4.15 | | 04-Ha | 1.78 | | 04-He | 1.78 | | 05-H | 2.62 (t-like ) | | 07-H | 6.14 | | 09-Ha | 3.42 | | 11-Ha | 1.75 | | 11-He | | | 12-Ha | | | 12-He | | | 15-Ha | | | 15-Hb | | | 16-Ha | 1.9 | | 16-Hb | 1.75 | | 17-H | 2.33 (t, 8.5) | | 18-Me | 1.07 | | 19-Me | 1.26 | | 21-Me | 1.43 | | 22-H | 3.71 | | 23-Ha | 1.31 | | 23-Hb | 1.66 | | 24-Ha | | | 24-Hb | | | 26-Me | 1.24 | | 27-Me | 1.24 |
| CD3OD | | 01-He | 3.82 | | 02-Ha | 3.87 (t, 3.1) | | 03-He | 4.04 | | 04-Ha | 1.80 (ddd, 13.4, 12.0, 4.8) | | 04-He | 1.75 | | 05-H | 2.61 (bdd, 12.0, 4.8) | | 07-H | 5.80 (bd, 2.6) | | 09-Ha | 3.08 (bd, 8.5) | | 11-Ha | 1.79 | | 11-He | 1.70 | | 12-Ha | 2.11 (td, 12.8, 12.8, 5.1) | | 12-He | 1.87 | | 15-Ha | 2.00 | | 15-Hb | 1.60 | | 16-Ha | 1.97 | | 16-Hb | 1.73 | | 17-H | 2.39 | | 18-Me | 0.905 (s) | | 19-Me | 0.905 (s) | | 20-H | | | 21-Me | 1.19 (s) | | 22-H | 3.32 (dd, 11.0, 1.8) | | 23-Ha | 1.66 | | 23-Hb | 1.29 | | 24-Ha | 1.78 | | 24-Hb | 1.43 | | 26-Me | 1.19 (s) | | 27-Me | 1.20 (s) |
|
| |
| M.P. | 244-246 °C | | [α]D20 | + 33.7 ? 2 ° (c 0.92; MeOH) | | IR (KBr) ν max (cm-1) | 3300-3500 (OH), 1605 (cyclohexenone) | | UV (EtOH) λ max (log ε) | 245 (4.09) |
| |
| TLC | NP-TLC (silica gel plates) Rf : 0.13 (EtOAc/MeOH/NH4OH 85:10:5), 0.12 (CH2Cl2/EtOH 85:15), 0.55 (EtOAc/EtOH/H2O 8:2:1), 0.39 (CHCl3/MeOH/Me2CO 6:2:1) (Rf 20E : respect-ively 0.18, 0.15, 0.61, 0.47); RP-TLC (on Merck C18 bonded plates) Rf : 0.45 (MeOH/H2O 50:50) (20E : 0.44) | | GLC | | | HPLC | 1- Retention time 20.0 min [Zorbax-Sil 250 mm x 4.6 mm i.d., solvent CH2Cl2/iPrOH/H2O 125:40:3 v/v/v, flow-rate 1 ml.min-1] (20E : 16.8 min); 2-Retention time 48.4 min [ Zorbax-Sil 250 mm x 4.6 mm i.d., solvent CH2Cl2/iPrOH/H2O 125:25:2 v/v/v, flow-rate 1 ml.min-1] (20E 37.4 min); 3-Retention time 4.6 min [ Spherisorb-5ODS2 250 mm x 4.6 mm i.d., solvent ACN- 0.1% TFA in H2O 23:77 /v/v, flow-rate 1 ml.min-1] (20E 5.5 min) |
| |
Drosophila melanogaster BII cell assay: EC50 = 2.0 x 10-7M | Galleria mellonella in vivo assay: ED50 = 15.6 ug/g | Sarcophaga bullata in vivo assay: ED50 = 15.6 ug/g |
| |
| First isolation | BALTAYEV, U.A. et al. (1977) Khim. Prir. Soedin. 813-819 |
 | | General | KHOLODOVA, YU. D. et al. (1979) Khim. Prir. Soedin. 171-174 |
 | | General | WILSON, I.D. et al. (1988) J. Planar Chromatogr. 1, 116-122 |
 | | General | GIRAULT, J.-P. et al. (1990) J. Nat. Prod. 53, 279-293 |
 | | Bioactivities | SLÁMA, K. et al. (1993) Insect Biochem. Molec. Biol. 23, 181-185 |
 | | Identification | VOKÁČ, K. et al. (2002) Collect. Czech. Chem. Commun. 67, 124-139 |
 | | Bioactivities | DINAN, L. et al. (2005) In: Comprehensive Molecular Insect Science (eds. L. I. Gilbert, C. Iatrou, S. Gill), Elsevier, Oxford, III, 197-242 |
 | | General | SIMON, A. et al. (2007) Steroids 72, 751-755 |
 |
| |
Permanent link to this datasheet: INTEGRISTERONE A
|
|




![Wikipedia: Rhaponticum integrifolium [Asteraceae]](/images/wikipedia.png)

