|
|
|
|
Year of first isolation: |
1974 |
| Formula: | C27H44O7 |
| Molecular weight: | 480 |
| Occurence in plants: |
Leuzea carthamoides [Asteraceae] »  ![Wikipedia: Leuzea carthamoides [Asteraceae]](/images/wikipedia.png)
Serratula coronata [Asteraceae] »  ![Wikipedia: Serratula coronata [Asteraceae]](/images/wikipedia.png)
Serratula wolffii [Asteraceae] »  ![Wikipedia: Serratula wolffii [Asteraceae]](/images/wikipedia.png)
|
| Occurence in animals: |
Manduca sexta [Lepidoptera] »  ![Wikipedia: Manduca sexta [Lepidoptera]](/images/wikipedia.png)
Pieris brassicae [Lepidoptera] »  ![Wikipedia: Pieris brassicae [Lepidoptera]](/images/wikipedia.png)
|
|
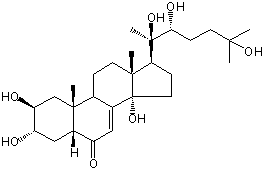
| |
| Canonical SMILES | CC12CCC3C(=CC(=O)C4C3(CC(C(C4)O)O)C)C1(CCC2C(C)(C(CCC(C)(C)O)O)O)O | Isomeric SMILES
[ PubChem: search | XML ]
[ ChemSpider: search ] | C1[C@]2([C@@H](C[C@@H]([C@H]1O)O)C(=O)C=C1[C@@H]2CC[C@]2([C@]1(CC[C@@H]2[C@](C)([C@@H](CCC(C)(C)O)O)O)O)C)C » 
| | IUPAC Name | (2S,3S,5R,9R,10R,13R,14S,17S)-2,3,14-trihydroxy-10,13-dimethyl-17-[(2R,3R)-2,3,6-trihydroxy-6-methylheptan-2-yl]-2,3,4,5,9,11,12,15,16,17-decahydro-1H-cyclopenta[a]phenanthren-6-one | | CAS-RN | | | PubChem CID | 70686927 | InChiKey
[ ChemIDPlus: search ] | NKDFYOWSKOHCCO-ZKSFWNCQSA-N | | InChI | InChI=1S/C27H44O7/c1-23(2,32)9-8-22(31)26(5,33)21-7-11-27(34)16-12-18(28)17-13-19(29)20(30)14-24(17,3)15(16)6-10-25(21,27)4/h12,15,17,19-22,29-34H,6-11,13-14H2,1-5H3/t15-,17-,19-,20-,21-,22+,24+,25+,26+,27+/m0/s1 |
| |
| EI-MS m/z (relative intensity %) | 480 [M]+, 462 (<1) [M-18]+, 444 (1), 426 (16), 411 (10), 408 (6), 393 (4), 375 (2), 363 (4), 352 (10), 345 (45), 344 (17), 329 (17), 328 (38), 327 (48), 313 (11), 311 (10), 310 (14), 309 (17), 301 (20); 300 (33), 285 (31), 269 (21), 267 (21), 173 (26), 99 (100) | | CI-MS (NH3) m/z | 481 [M+H]+, 463, 445, 427, 363, 345 | | FAB-MS | 503 (85) [M+Na]+, 481 (21) [M+H]+, 463 (36), 445 (100), 427 (98), 409 (36) |
|
|
| CD3OD | | 01 | 43.03 | | 02 | 72.11 | | 03 | 75.36 | | 04 | 33.65 | | 05 | 57.47 | | 06 | 204.65 | | 07 | 121.99 | | 08 | 168.12 | | 09 | 35.97 | | 10 | 39.61 | | 11 | 21.50 | | 12 | 32.48 | | 13 | ~49.0 | | 14 | 85.10 | | 15 | 31.74 | | 16 | 21.50 | | 17 | 50.55 | | 18 | 18.03 | | 19 | 23.88 | | 20 | 77.93 | | 21 | 21.04 | | 22 | 78.44 | | 23 | 27.35 | | 24 | 42.40 | | 25 | 71.31 | | 26 | 29.69 | | 27 | 28.96 |
| C5D5N | | 01 | 42.91 | | 02 | 71.62 | | 03 | 74.95 | | 04 | 33.48 | | 05 | 56.87 | | 06 | 201.44 | | 07 | 121.15 | | 08 | 165.84 | | 09 | 35.08 | | 10 | 38.67 | | 11 | 20.78 | | 12 | 31.67 | | 13 | 47.81 | | 14 | 83.80 | | 15 | 31.41 | | 16 | 21.19 | | 17 | 49.79 | | 18 | 17.58 | | 19 | 23.65 | | 20 | 76.54 | | 21 | 21.40 | | 22 | 77.26 | | 23 | 27.21 | | 24 | 42.36 | | 25 | 69.24 | | 26 | 29.74 | | 27 | 29.80 |
|
| CD3OD | | 01-Ha | 2.10 (dd 13.0, 11.6) | | 01-He | 1.09 (dd 13.0, 4.4) | | 02-Ha | 3.64 (ddd 11.6, 8.8, 4.4) | | 03-Ha | 3.35 (ddd 11.2, 8.8, 4.7) | | 04-Ha | 1.57 | | 04-He | 1.75 | | 05-H | 2.40 (dd 13.0, 4.5) | | 07-H | 5.82 (d 2.6) | | 09-Ha | 3.18 (ddd 11.9, 6.8, 2.6) | | 11-Ha | 1.70 | | 11-He | 1.82 | | 12-Ha | 2.14 | | 12-He | 1.88 | | 15-Ha | 1.96 | | 15-Hb | 1.61 | | 16-Ha | 1.74 | | 16-Hb | 1.99 | | 17-H | 2.40 | | 18-Me | 0.89 (s) | | 19-Me | 0.96 (s) | | 21-Me | 1.21 (s) | | 22-H | 3.33 (dd ~11.0, 2.0) | | 23-Ha | 1.66 | | 23-Hb | 1.29 | | 24-Ha | 1.80 | | 24-Hb | 1.43 | | 26-Me | 1.20 (s) | | 27-Me | 1.19 (s) |
| C5D5N | | 01-Ha | 2.45 (dd 12.9, 4.3) | | 01-He | 1.47 (dd 12.9, 11.0) | | 02-Ha | 4.24 (ddd 11.0, 8.8, 4.3) | | 03-Ha | 3.86 (m) | | 04-Ha | 1.95 (m) | | 04-He | 2.17 (m) | | 05-H | 2.47 (m) | | 07-H | 6.24 (d 2.1) | | 09-Ha | 3.71 (ddd 8.8, 4.7, 2.1) | | 11-Ha | 1.84 | | 11-He | 1.76 | | 12-Ha | 2.54 (ddd 12.9, 12.8, 4.9) | | 12-He | 2.02 (m) | | 15-Ha | 2.17 (m) | | 15-Hb | 1.88 (m) | | 16-Ha | 2.09 (m) | | 16-Hb | 2.45 (m) | | 17-H | 3.00 (m) | | 18-Me | 1.23 (s) | | 19-Me | 1.06 (s) | | 21-Me | 1.59 (s) | | 22-H | 3.88 (m) | | 23-Ha | 2.15 (m) | | 23-Hb | 1.83 (m) | | 24-Ha | 2.28 (m) | | 24-Hb | 1.74 (m) | | 26-Me | 1.39 (s) | | 27-Me | 1.39 (s) |
|
| |
| M.P. | 225-229 °C ; | | [α]D20 | +61.5 (c 0.88, MeOH) | | IR (KBr) ν max (cm-1) | 3410, 1653 | | UV (EtOH) λ max (log ε) | 245 (4.033); |
| |
| TLC on silica | Rf 0.17 (CHCl3-EtOH 4:1) or 0.34 (if continuous development for 2.5 hours) | | HPLC | NP-HPLC on APS hypersil (25 cm x 4.6 mm i.d.), Retention volume 184 ml (CH2Cl2-MeOH-iPrOH 95:1:4 v/v/v) (20E : 87 ml); Retention volume 110 ml (CH2Cl2-MeOH-iPrOH 95:2:3 v/v/v) (20E : 65 ml); Retention volume 52 ml (CH2Cl2-MeOH-iPrOH 95:3:2 v/v/v) (20E : 42 ml). | | NP-HPLC | | |
| N o t i c e: The first isolation of 3-epi-20E from a plant species, described in 2000 (by Odinokov et al., Rus. Chem. Bull., Int. Ed., 49/11, 1923-4 as CORONATASTERONE was presented with incorrect structure, and later in 2002 was revised (see Odinokov et al., in References). | | IS-RP-HPLC | |
| |
Drosophila melanogaster BII
cell assay: EC50 = 1.3 x 10-7M | Drosophila melanogaster BII
cell assay: EC50 = 1.6 x 10-7M | Musca assay: 7-10%
(20-hydroxyecdysone = 100%) |
| |
| First isolation | THOMPSON, M.J. et al. (1974) Steroids 24, 359-366 |
 | | General | DINAN, L. et al. (1978) Steroids 32, 629-638 |
 | | Bioactivities | BERGAMASCO, R. et al. (1980) In: Progress in Ecdysone Research (Ed. J.A. Hoffmann), Elsevier/North Holland Biomed. Press, Amsterdam 299-324 |
 | | General | DINAN, L. et al. (1981) J. Chromatogr. 205, 139-145 |
 | | General | BLAIS, C. et al. (1984) Hoppe-Seyler´s Z. Physiol. Chem. 365, 809-817 |
 | | General | BEYDON, P. et al. (1987) Aust. J. Chem. 4, 139-149 |
 | | General | THOMPSON, M.J. et al. (1989) In: Ecdysone, from chemistry to mode of action (J. Koolman Ed.), Georg Thieme Verlag, Stuttgart, pp. 65-73 |
 | | Bioactivities | RAVI, M. et al. (2001) J. Chem. Inf. Comput. Sci. 41, 1587-1604 |
 | | First isolation | ODINOKOV, V.N. et al. (2002) Insect Biochem. Molec. Biol. 32, 161-165 |
 | | Identification | VOKÁČ, K. et al. (2002) Collect. Czech. Chem. Commun. 67, 124-139 |
 | | General | HUNYADI, A. et al. (2007) J. Chrom. Sci. 45, 76-86 |
 | | General | HUNYADI, A. et al. (2008) Chromatographia 67, 767-772 |
 |
| |
Permanent link to this datasheet: 3-EPI-20-HYDROXYECDYSONE
|
|




![Wikipedia: Leuzea carthamoides [Asteraceae]](/images/wikipedia.png)

