|
|
|
|
Year of first isolation: |
1967 |
| Formula: | C27H44O7 |
| Molecular weight: | 480 |
| Occurence in plants: |
Thelypteris palustris [Thelypteridaceae] »  ![Wikipedia: Thelypteris palustris [Thelypteridaceae]](/images/wikipedia.png)
Onoclea sensibilis [Onocleaceae] »  ![Wikipedia: Onoclea sensibilis [Onocleaceae]](/images/wikipedia.png)
Pfaffia iresinoides [Amaranthaceae] »  ![Wikipedia: Pfaffia iresinoides [Amaranthaceae]](/images/wikipedia.png)
Pfaffia paniculata [Amaranthaceae] »  ![Wikipedia: Pfaffia paniculata [Amaranthaceae]](/images/wikipedia.png)
Polypodium vulgare [Polypodiaceae] »  ![Wikipedia: Polypodium vulgare [Polypodiaceae]](/images/wikipedia.png)
|
| Occurence in animals: |
|
|
|
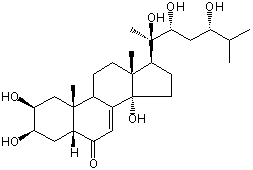
| |
| Canonical SMILES | CC(C)C(CC(C(C)(C1CCC2(C1(CCC3C2=CC(=O)C4C3(CC(C(C4)O)O)C)C)O)O)O)O | Isomeric SMILES
[ PubChem: search | XML ]
[ ChemSpider: search ] | C1[C@]2([C@@H](C[C@H]([C@H]1O)O)C(=O)C=C1[C@@H]2CC[C@]2([C@]1(CC[C@@H]2[C@](C)([C@H](O)C[C@@H](C(C)C)O)O)O)C)C » 
| | IUPAC Name | (2S,3R,5R,9R,10R,13R,14S,17S)-2,3,14-trihydroxy-10,13-dimethyl-17-[(2R,3R,5S)-2,3,5-trihydroxy-6-methylheptan-2-yl]-2,3,4,5,9,11,12,15,16,17-decahydro-1H-cyclopenta[a]phenanthren-6-one | | CAS-RN | 18089-44-6 | | PubChem CID | 441836 | InChiKey
[ ChemIDPlus: search ] | UMMBJCYNGLCGEF-OAUIFJKNSA-N | | InChI | InChI=1S/C27H44O7/c1-14(2)18(28)12-23(32)26(5,33)22-7-9-27(34)16-10-19(29)17-11-20(30)21(31)13-24(17,3)15(16)6-8-25(22,27)4/h10,14-15,17-18,20-23,28,30-34H,6-9,11-13H2,1-5H3/t15-,17-,18-,20+,21-,22-,23+,24+,25+,26+,27+/m0/s1 |
| |
| CI-MS (NH3) m/z | | | EI-MS m/z (relative intensity %) | 480, 462, 444, 426, 408, 363 (M-117), 345, 328, 117, 99, 81 | | HR-MS | |
|
|
| C5D5N | | 01 | 37.7 | | 02 | 68.1 | | 03 | 68.0 | | 04 | 32.4 | | 05 | 51.3 | | 06 | 203.8 | | 07 | 121.6 | | 08 | 166.3 | | 09 | 34.3 | | 10 | 38.6 | | 11 | 21.0 | | 12 | 31.9 | | 13 | 48.0 | | 14 | 84.1 | | 15 | 31.6 | | 16 | 21.4 | | 17 | 49.9 | | 18 | 17.9 | | 19 | 24.4 | | 20 | 76.8 | | 21 | 21.6 | | 22 | 77.4 | | 23 | 35.7 | | 24 | 76.7 | | 25 | 33.8 | | 26 | 17.0 | | 27 | 19.5 |
|
| C5D5N | | 01-Ha | | | 01-Hb | | | 02-Ha | 4.16 (br dt, 11.8, 3.4) | | 03-He | 4.22 (br s ) | | 04-Ha | | | 04-He | | | 05-H | 3.02 (dd, 13.0, 4.3) | | 07-H | 6.25 (d, 2.2) | | 09-Ha | 3.58 (br t, 9.2) | | 11-Ha | | | 11-He | | | 12-Ha | 2.60 (dt, 13.0, 5.0) | | 12-He | | | 15-Ha | | | 15-Hb | | | 16-Ha | 2.47 (q, 10.4) | | 16-Hb | | | 17-H | 2.94 (t, 9.2) | | 18-Me | 1.21 (s ) 1.18* | | 19-Me | 1.06 (s ) 1.05* | | 21-Me | 1.59 (s ) 1.54* | | 22-H | 4.12 (br d, 10.6) | | 23-Ha | | | 23-Hb | | | 24-H | 3.94 (dt, 9.0, 4.0) | | 26-Me | 1.003 (d, 6.6) 1.00* | | 27-Me | 1.006 (d, 6.6) 1.00* |
|
| |
| M.P. | 229-230 °C | | [α]D20 | + 7.4° (c ; MeOH) | | IR (KBr) ν max (cm-1) | 3420 (OH), 1650 (cyclohexenone) | | UV (EtOH) λ max (log ε) | 243 (?) |
| |
| HPTLC | | | TLC | silicagel plates, Rf 0.54 (CHCl3-MeOH-H2O, 13:7:2) | | GLC | | | HPLC | |
| |
Drosophila melanogaster BII cell assay: EC50 = 2.0 x 10-9M | Sarcophaga assay: highly active |
| |
| First isolation | TAKEMOTO, T. et al. (1967) Chem. Pharm. Bull. 15, 1816 |
 | | General | TAKEMOTO, T. et al. (1968) Tetrahedron Lett. 375-378 |
 | | General | BLUNT, J.W. et al. (1979) Aust. J. Chem. 32, 779-782 |
 | | Bioactivities | BERGAMASCO, R. et al. (1980) In: Progress in Ecdysone Research (Ed. J.A. Hoffmann), Elsevier/North Holland Biomed. Press, Amsterdam 299-324 |
 | | General | NISHIMOTO, N. et al. (1988) Phytochemistry 27, 1665-1668 |
 | | General | NISHIMOTO, N. et al. (1988) Phytochemistry 26, 2505-2507 |
 | | Identification | COLL, J. et al. (1994) Tetrahedron 50, 7247-7252 |
 | | Bioactivities | DINAN, L. et al. (2005) In: Comprehensive Molecular Insect Science (eds. L. I. Gilbert, C. Iatrou, S. Gill), Elsevier, Oxford, III, 197-242 |
 | | General | LI, J. et al. (2010) Planta Med. 76(6): 635-639 |
 |
| |
Permanent link to this datasheet: PTEROSTERONE
|
|




![Wikipedia: Thelypteris palustris [Thelypteridaceae]](/images/wikipedia.png)

