|
|
|
|
Year of first isolation: |
1978 |
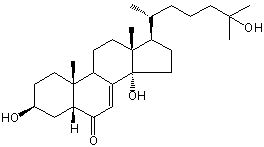
| Formula: | C27H44O4 |
| Molecular weight: | 432 |
| Occurence in plants: |
|
|
| Occurence in animals: |
Locusta migratoria [Orthoptera] »  ![Wikipedia: Locusta migratoria [Orthoptera]](/images/wikipedia.png)
|
|
| |
| Canonical SMILES | CC(CCCC(C)(C)O)C1CCC2(C1(CCC3C2=CC(=O)C4C3(CCC(C4)O)C)C)O | Isomeric SMILES
[ PubChem: search | XML ]
[ ChemSpider: search ] | C1[C@]2([C@@H](C[C@H](C1)O)C(=O)C=C1[C@@H]2CC[C@]2([C@]1(CC[C@@H]2[C@H](C)CCCC(C)(C)O)O)C)C » 
| | IUPAC Name | (3S,5R,9R,10R,13R,14S,17R)-3,14-dihydroxy-17-[(2R)-6-hydroxy-6-methylheptan-2-yl]-10,13-dimethyl-2,3,4,5,9,11,12,15,16,17-decahydro-1H-cyclopenta[a]phenanthren-6-one | | CAS-RN | | | PubChem CID | 14017685 | InChiKey
[ ChemIDPlus: search ] | CRCTVUFFBCIURJ-DJHAZVFJSA-N | | InChI | InChI=1S/C27H44O4/c1-17(7-6-11-24(2,3)30)19-10-14-27(31)21-16-23(29)22-15-18(28)8-12-25(22,4)20(21)9-13-26(19,27)5/h16-20,22,28,30-31H,6-15H2,1-5H3/t17-,18+,19-,20+,22+,25-,26-,27-/m1/s1 |
| |
| GC-MS m/z (relative intensity %) - Trisilylated compound | 648 (M+, C36H68O4Si3) (22), 633 (21), 620 (39), 558 (23), 543 (17), 468 (20), 377 (13), 131 (100). | | EI-MS m/z (relative intensity %) | 432 (M+, C27H44O4) (10); 414 (21), 404 (29), 399 (29), 396(10), 386 (90), 381 (10), 372 (6), 353 (13), 341 (4), 325 (3), 323 (2), 315 (9), 285 (11), 234 (49), 233 (100), 215 (16), 207 (15), 194 (11). |
|
|
| CD2Cl2 | | 01 | 26.8 * | | 02 | 29.9 * | | 03 | 65.0 | | 04 | 31.9 * | | 05 | 50.8 $ | | 06 | 203.1 | | 07 | 121.4 | | 08 | 164.6 | | 09 | 34.4 | | 10 | | | 11 | 21.1 | | 12 | 32.6 * | | 13 | 46.3 | | 14 | 85.6 | | 15 | 31.3 * | | 16 | 28.5 * | | 17 | 50.8 $ | | 18 | 15.9 | | 19 | 23.9 | | 20 | 35.8 | | 21 | 18.8 | | 22 | 36.8 | | 23 | 21.1 | | 24 | 44.6 | | 25 | 70.9 | | 26 | 29.4 | | 27 | 29.2 |
|
| CD3OD | | 01-Ha | | | 01-He | | | 02-Ha | | | 03-He | 4.04 | | 04-Ha | | | 04-He | | | 05-H | 2.41 | | 07-H | 5.83 | | 09-Ha | 3.10 | | 11-Ha | | | 11-He | | | 12-Ha | | | 12-He | | | 14-H | | | 15-Ha | | | 15-Hb | | | 16-Ha | | | 16-Hb | | | 17-H | | | 18-Me | 0.72 | | 19-Me | 1.00 | | 21-Me | 0.97 | | 22-H | | | 23-Ha | | | 23-Hb | | | 24-Ha | | | 24-Hb | | | 25-H | | | 26-Me | 1.21 | | 27-Me | 1.21 |
|
| |
| M.P. | 262-263 °C | | [α]D21 | + 63° (c 5.9; MeOH). | | IR (KBr) ν max (cm-1) | 3600, 2970, 2940, 2880, 1660, 1600, 1440-1470, 1380, 1150, 1 | | UV (EtOH) λ max (log ε) | 241 (4.061) |
| |
| HPTLC | | | TLC | Rf 0.3 (silica plates, solvent Toluene-Me2CO-Et2MeN 6: 4: 0.1 v/v/v) | | GLC | | | HPLC | RP : column Spherisorb 5-ODS-2, solvent linear gradient (in 30 min) of 20% to 100% ACN-iPrOH (5:2) in 0.1% TFA in water, flow-rate 1 ml.min-1, Rt 25.5 min (2dE : 17.0 min) |
| |
| |
| |
Permanent link to this datasheet: 2,22-DIDEOXYECDYSONE
|
|




![Wikipedia: Locusta migratoria [Orthoptera]](/images/wikipedia.png)

