|
|
|
|
Year of first isolation: |
1995 |
| Formula: | C31H46O9 |
| Molecular weight: | 562 |
| Occurence in plants: |
Ajuga reptans var. atropurpurea [Lamiaceae (alt. Labiatae)] »  ![Wikipedia: Ajuga reptans var. atropurpurea [Lamiaceae (alt. Labiatae)]](/images/wikipedia.png)
|
| Occurence in animals: |
|
|
|
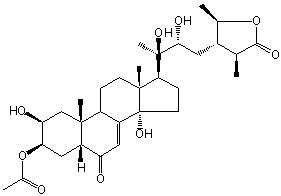
| |
| Canonical SMILES | | Isomeric SMILES
[ PubChem: search | XML ]
[ ChemSpider: search ] | C1[C@]2([C@@H](C[C@H]([C@H]1O)OC(=O)C)C(=O)C=C1[C@@H]2CC[C@]2([C@]1(CC[C@@H]2[C@](C)([C@@H](C[C@@H]1[C@H](OC(=O)[C@H]1C)C)O)O)O)C)C » 
| | IUPAC Name | (2S,3R,5R,9R,10R,13R,14S,17S)-17-((2R,3R)-4-((2R,3S,4S)-2,4-dimethyl-5-oxotetrahydrofuran-3-yl)-2,3-dihydroxybutan-2-yl)-2,14-dihydroxy-10,13-dimethyl-6-oxo-2,3,4,5,6,9,10,11,12,13,14,15,16,17-tetradecahydro-1H-cyclopenta[a]phenanthren-3-yl acetate | | CAS-RN | | | PubChem CID | | InChiKey
[ ChemIDPlus: search ] | WXZKWXBGILJBMI-UVORWOJQSA-N | | InChI | InChI=1S/C31H46O9/c1-15-18(16(2)39-27(15)36)11-26(35)30(6,37)25-8-10-31(38)20-12-22(33)21-13-24(40-17(3)32)23(34)14-28(21,4)19(20)7-9-29(25,31)5/h12,15-16,18-19,21,23-26,34-35,37-38H,7-11,13-14H2,1-6H3/t15-,16+,18-,19-,21-,23-,24+,25-,26+,28+,29+,30+,31+/m0/s1 |
| |
| CI-MS (NH3) m/z | | | MS (thermospray) m/z | 580 [M+NH4]+, 563 [M+H]+, 562 [M]+, 545 [M+H-H2O]+, 520 [M+NH4-CH3COOH]+, 502 [M-CH3COOH]+ | | HR-MS | |
|
|
| C5D5N | | 01 | 38.74 * | | 02 | 66.10 | | 03 | 71.29 | | 04 | 29.65 | | 05 | 51.95 | | 06 | 202.13 | | 07 | 121.65 | | 08 | 166.21 | | 09 | 34.41 | | 10 | 38.62 * | | 11 | 21.09 & | | 12 | 31.91 | | 13 | 48.17 | | 14 | 84.11 | | 15 | 32.01 | | 16 | 21.36 & | | 17 | 49.66 | | 18 | 17.91 | | 19 | 24.28 | | 20 | 76.77 | | 21 | 21.01 | | 22 | 73.99 | | 23 | 34.52 | | 24 | 48.70 | | 25 | 42.46 | | 26 | 179.18 | | 27 | 15.94 | | 28 | 79.83 | | 29 | 19.35 | | CH3CO- | 21.09, 170.57 |
|
| C5D5N | | 01-Ha | | | 01-Hb | | | 02-Ha | 4.29 (br, w1/2 = 22.3) | | 03-He | 5.50 (br, w1/2 = 9.3) | | 04-Ha | | | 04-He | | | 05-H | 2.67 (dd, 13.2, 4.2) | | 07-H | 6.28 (d, 2.4) | | 09-Ha | 3.59 (brt, w1/2 = 23.0) | | 11-Ha | | | 11-He | | | 12-Ha | | | 12-He | | | 15-Ha | | | 15-Hb | | | 16-Ha | | | 16-Hb | | | 17-H | 2.87 (bt, 9.0) | | 18-Me | 1.25 (s ) | | 19-Me | 1.09 (s ) | | 21-Me | 1.58 (s ) | | 22-H | 3.95 (br, w1/2 = 17) | | 23-Ha | | | 23-Hb | | | 24-H | | | 25-H | 2.38 (dq, 11.1, 6.9) | | 26-Me | ? | | 27-Me | 1.37 (d, 6.9) | | 28-H | 4.01 (dq, 9.3, 6.0) | | 29-Me | 1.31 (d, 6) | | Ac-O | 1.97 (s ) |
|
| |
| M.P. | | | [α]D20 | | | IR (KBr) ν max (cm-1) | | | UV (EtOH) λ max (log ε) | |
| |
| HPTLC | | | TLC | | | GLC | | | HPLC | RP Lichrospher 100 RP-18, 5 ?m, solvent iPrOH-H2O 1:10, 55°C, 1 ml.min-1, Retention time 20 min (coelutes with 20E3Ac); RP Spherisorb 10ODS2, 300 x 7.8 mm, solvent iPrOH-H2O 1.1:10, 55°C, 3 ml.min-1 Retention time 51.2 min (vs 39.1 min for 29NCy3Ac) |
| |
Drosophila melanogaster BII cell assay: EC50 = 4.3 x 10-7M |
| |
| |
Permanent link to this datasheet: CYASTERONE 3-ACETATE
|
|




![Wikipedia: Ajuga reptans var. atropurpurea [Lamiaceae (alt. Labiatae)]](/images/wikipedia.png)

