|
|
|
|
Year of first isolation: |
1982 |
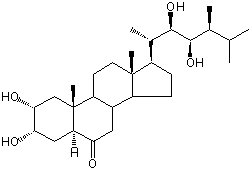
| Formula: | C28H48O5 |
| Molecular weight: | 464 |
| Occurence in plants: |
Castanea (chestnut tree) insect galls [Fagaceae] »  ![Wikipedia: Castanea (chestnut tree) insect galls [Fagaceae]](/images/wikipedia.png)
|
| Occurence in animals: |
|
|
|
| |
| Canonical SMILES | CC(C)C(C)C(C(C(C)C1CCC2C1(CCC3C2CC(=O)C4C3(CC(C(C4)O)O)C)C)O)O | Isomeric SMILES
[ PubChem: search | XML ]
[ ChemSpider: search ] | C1[C@]2([C@H](C[C@@H]([C@@H]1O)O)C(=O)CC1[C@@H]2CC[C@]2(C1CC[C@@H]2[C@H](C)[C@H]([C@@H](C(C(C)C)C)O)O)C)C » 
| | IUPAC Name | (2R,3S,5S,8S,9S,10R,13S,14S,17R)-17-[(2S,3R,4R,5S)-3,4-dihydroxy-5,6-dimethylheptan-2-yl]-2,3-dihydroxy-10,13-dimethyl-1,2,3,4,5,7,8,9,11,12,14,15,16,17-tetradecahydrocyclopenta[a]phenanthren-6-one | | CAS-RN | 80736-41-0 | | PubChem CID | 133534 | InChiKey
[ ChemIDPlus: search ] | VYUIKSFYFRVQLF-YLNAYWRASA-N | | InChI | nChI=1S/C28H48O5/c1-14(2)15(3)25(32)26(33)16(4)18-7-8-19-17-11-22(29)21-12-23(30)24(31)13-28(21,6)20(17)9-10-27(18,19)5/h14-21,23-26,30-33H,7-13H2,1-6H3/t15-,16-,17-,18+,19-,20-,21+,23-,24+,25+,26+,27+,28+/m0/s1 |
| |
| EI-MS m/z (relative intensity %) | 403 (0.4), 385 (0.3), 393 (3), 375 (2), 357 (2), 364 (100), 363 (35), 362 (36), 346 (42), 345 (59), 327 (36), 315 (7), 305 (5), 287 (17), 269 (10), 263 (11), 245 (13), 227 (5), 155 (5). | | HR-MS | | | FAB-MS m/z | 465 (M+1)+, 447 (M+1-18), 429, 393, 364 |
|
|
| C5D5N | | 01 | | | 02 | | | 03 | | | 04 | | | 05 | | | 06 | | | 07 | | | 08 | | | 09 | | | 10 | | | 11 | | | 12 | | | 13 | | | 14 | | | 15 | | | 16 | | | 17 | | | 18 | | | 19 | | | 20 | | | 21 | | | 22 | | | 23 | | | 24 | | | 25 | | | 26 | | | 27 | | | 28 | |
|
| CDCl3 | | 01-Ha | | | 01-Hb | | | 02-H | 3.77 (br, w1/2=23) | | 03-H | 4.06 (br, w1/2= 8) | | 04-Ha | | | 04-He | | | 05-H | 2.69 (dd, 13, 4) | | 07-Ha | | | 07-Hb | | | 09-Ha | | | 11-Ha | | | 11-He | | | 12-Ha | | | 12-He | | | 14-H | | | 15-Ha | | | 15-Hb | | | 16-Ha | | | 16-Hb | | | 17-H | | | 18-Me | 0.69 (s ) | | 19-Me | 0.76 (s ) | | 22-H | 3.56 (d, 9)* | | 23-H | 3.72 (d, 9)* | | 24-Ha | | | 24-Hb | | | 25-H | | | Me' | 0.85 (d ) | | Me'' | 0.91 (d ) | | Me''' | 0.95 (d ) | | Me'''' | 0.97 (d ) |
|
| |
| M.P. | 259-261°C | | [α]D20 | | | IR (KBr) ν max (cm-1) | | | UV (EtOH) λ max (log ε) | |
| |
| HPTLC | | | TLC on silica | Rf 0.38 (CHCl3-EtOH 6:1) | | GLC | GLC-MS of bismethane-boronate | | HPLC | NP-HPLC, Partisil 5 (CHCl3-iPrOH, gradient); RP-HPLC, Develosil ODS-3 (ACN-H2O 1:1) |
| |
Drosophila melanogaster BII cell assay: EC50 = >10-3M |
| |
| |
Permanent link to this datasheet: CASTASTERONE
|
|




![Wikipedia: Castanea (chestnut tree) insect galls [Fagaceae]](/images/wikipedia.png)

