|
|
|
|
Year of first isolation: |
2023 |
| Formula: | C27H42O5 |
| Molecular weight: | 446 |
| Occurence in plants: |
Dianthus superbus [Caryophyllaceae] »  ![Wikipedia: Dianthus superbus [Caryophyllaceae]](/images/wikipedia.png)
|
| Occurence in animals: |
|
|
|
| |
Isomeric SMILES
[ PubChem: search | XML ]
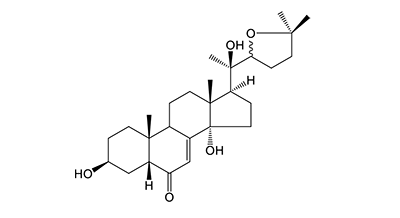
[ ChemSpider: search ] | C[C@@]12[C@@](C(C=C3C2CC[C@@]4(C)[C@@]3(O)CC[C@]4([H])[C@](C)(C5CCC(C)(C)O5)O)=O)([H])C[C@@H](O)CC1 » 
| | IUPAC Name | (3S,5R,10R,13R,14S,17S)-17-((1R)-1-(5,5-dimethyltetrahydrofuran-2-yl)-1-hydroxyethyl)-3,14-dihydroxy-10,13-dimethyl-1,2,3,4,5,9,10,11,12,13,14,15,16,17-tetradecahydro-6H-cyclopenta[a]phenanthren-6-one | | CAS-RN | | | PubChem CID | | InChiKey
[ ChemIDPlus: search ] | UHPHPHNUELAFCV-SYUIPJMMSA-N | | InChI | InChI=1S/C27H42O5/c1-23(2)10-9-22(32-23)26(5,30)21-8-13-27(31)18-15-20(29)19-14-16(28)6-11-24(19,3)17(18)7-12-25(21,27)4/h15-17,19,21-22,28,30-31H,6-14H2,1-5H3/t16-,17?,19-,21-,22?,24+,25+,26+,27+/m0/s1 |
| |
| HRESIMS (positive ion mode) m/z
| 469.2922 [M+Na]+ , calculated for C27H42O4Na+ 469.2930 |
|
|
| CD3OD | | 01 | 28.2 | | 02 | 27.5 | | 03 | 64.1 | | 04 | 31.9 | | 05 | 51.1 | | 06 | 205.9 | | 07 | 120.6 | | 08 | 167.1 | | 09 | 33.0 | | 10 | 36.2 | | 11 | 20.4 | | 12 | 31.1 | | 13 | 47.4 | | 14 | 84.0 | | 15 | 30.2 | | 16 | 20.4 | | 17 | 50.4 | | 18 | 16.7 | | 19 | 23.0 | | 20 | 75.6 | | 21 | 19.3 | | 22 | 84.1 | | 23 | 27.0 | | 24 | 38.2 | | 25 | 80.4 | | 26 | 27.6 | | 27 | 27.0 |
|
| CD3OD | | 01-Ha | 1.49 (m) | | 01-Hb | 1.62 (m) | | 02-Ha | 1.67 (m) | | 02-He | 1.80 (m) | | 03-He | 4.01 (br, s) | | 04-Ha | 1.80 (m) | | 04-Hb | 1.58 (m) | | 05-H | 2.44 (dd, 12.4,4.0) | | 07-H | 5.82 (d, 2.2) | | 09-H | 3.22 (m) | | 11-Ha | 1.74 (m) | | 11-Hb | 1.66 (m) | | 12-Ha | 2.15 (m) | | 12-Hb | 1.85 (m) | | 15-Ha | 1.99 (m) | | 15-Hb | 1.60 (m) | | 16-Ha | 2.01 (m) | | 16-Hb | 1.85 (m) | | 17-H | 2.40 (m) | | 18-Me | 0.86 (s) | | 19-Me | 0.98 (s) | | 20-H | - | | 21-Me | 1.23 (s) | | 22-H | 3.94 (m) | | 23-Ha | 1.92 (m) | | 23-Hb | 1.77 (m) | | 24-Ha | 1.77 (m) | | 24-Hb | 1.72 (m) | | 26-Me | 1.26 (s) | | 27-Me | 1.27 (s) |
|
| |
| M.P. | — °C ; | | [α]D20 | ° (c ; MeOH) | | IR (KBr) ν max (cm-1) | | | UV (MeOH) λ max (log ε) | - nm (-) ; |
| |
| |
| |
| |
Permanent link to this datasheet: SUPERECDYSONE B
|
|




![Wikipedia: Dianthus superbus [Caryophyllaceae]](/images/wikipedia.png)

