|
|
|
|
Year of first isolation: |
1968 |
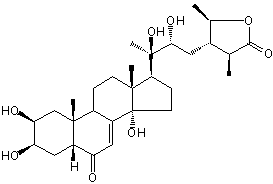
| Formula: | C29H44O8 |
| Molecular weight: | 520 |
| Occurence in plants: |
Ajuga nipponensis [Lamiaceae (alt. Labiatae)] »  ![Wikipedia: Ajuga nipponensis [Lamiaceae (alt. Labiatae)]](/images/wikipedia.png)
Cyathula capitata [Amaranthaceae] »  ![Wikipedia: Cyathula capitata [Amaranthaceae]](/images/wikipedia.png)
Ajuga chia [Lamiaceae (alt. Labiatae)] »  ![Wikipedia: Ajuga chia [Lamiaceae (alt. Labiatae)]](/images/wikipedia.png)
Ajuga decumbens [Lamiaceae (alt. Labiatae)] »  ![Wikipedia: Ajuga decumbens [Lamiaceae (alt. Labiatae)]](/images/wikipedia.png)
Ajuga incisa [Lamiaceae (alt. Labiatae)] »  ![Wikipedia: Ajuga incisa [Lamiaceae (alt. Labiatae)]](/images/wikipedia.png)
Ajuga remota [Lamiaceae (alt. Labiatae)] »  ![Wikipedia: Ajuga remota [Lamiaceae (alt. Labiatae)]](/images/wikipedia.png)
Ajuga reptans [Lamiaceae (alt. Labiatae)] »  ![Wikipedia: Ajuga reptans [Lamiaceae (alt. Labiatae)]](/images/wikipedia.png)
|
| Occurence in animals: |
|
|
|
| |
| Canonical SMILES | CC1C(C(OC1=O)C)CC(C(C)(C2CCC3(C2(CCC4C3=CC(=O)C5C4(CC(C(C5)O)O)C)C)O)O)O | Isomeric SMILES
[ PubChem: search | XML ]
[ ChemSpider: search ] | C1[C@]2([C@@H](C[C@H]([C@H]1O)O)C(=O)C=C1[C@@H]2CC[C@]2([C@]1(CC[C@@H]2[C@](C)([C@@H](C[C@@H]1[C@H](OC(=O)[C@H]1C)C)O)O)O)C)C » 
| | IUPAC Name | (3S,4S,5R)-4-[(2R,3R)-2,3-dihydroxy-3-[(2S,3R,5R,9R,10R,13R,14S,17S)-2,3,14-trihydroxy-10,13-dimethyl-6-oxo-2,3,4,5,9,11,12,15,16,17-decahydro-1H-cyclopenta[a]phenanthren-17-yl]butyl]-3,5-dimethyloxolan-2-one | | CAS-RN | 17086-76-9 | | PubChem CID | 119444 | InChiKey
[ ChemIDPlus: search ] | NEFYSBQJYCICOG-YSEUJXISSA-N | | InChI | InChI=1S/C29H44O8/c1-14-16(15(2)37-25(14)34)10-24(33)28(5,35)23-7-9-29(36)18-11-20(30)19-12-21(31)22(32)13-26(19,3)17(18)6-8-27(23,29)4/h11,14-17,19,21-24,31-33,35-36H,6-10,12-13H2,1-5H3/t14-,15+,16-,17-,19-,21+,22-,23-,24+,26+,27+,28+,29+/m0/s1 |
| |
| CI-MS (NH3) m/z | | | EI-MS m/z | 520 [M]+, 505 [M-15]+, 502 [M-18]+, 484, 466, 451, 448, 433, 415, 363, 345, 327, 300, 201, 183, 157, 43 | | HR-MS | | | EI-MS m/z (relative intensity %) | 520 (0.4), 505 (0.2), 502 (6), 487 (1), 484 (16), 469 (7), 466 (13), 451 (7), 448 (4), 433 (8), 430 (1), 363 (7), 345 (29), 327 (30), 300 (17), 183 (28). |
|
|
| CDCl3 | | 01 | 37.9 | | 02 | 68.0 | | 03 | 68.0 | | 04 | 32.0 * | | 05 | 51.3 | | 06 | 201.0 | | 07 | 122.2 | | 08 | 166.1 | | 09 | 34.8 | | 10 | 38.6 | | 11 | 21.3 | | 12 | 31.8 * | | 13 | 48.7 | | 14 | 84.1 | | 15 | 32.4 * | | 16 | 21.0 ** | | 17 | 50.0 | | 18 | 17.8 | | 19 | 24.4 | | 20 | 76.7 | | 21 | 21.1 ** | | 22 | 73.9 | | 23 | 34.4 | | 24 | 48.1 | | 25 | 42.2 | | 26 | 179.3 | | 27 | 15.8 | | 28 | 79.8 | | 29 | 19.3 |
| C5D5N | | 01 | 37.97 | | 02 | 68.10 | | 03 | 68.03 | | 04 | 32.46 | | 05 | 51.39 | | 06 | 203.44 | | 07 | 121.84 | | 08 | 165.84 | | 09 | 34.48 | | 10 | 38.70 | | 11 | 21.09 | | 12 | 32.04 | | 13 | 48.18 | | 14 | 84.13 | | 15 | 31.92 | | 16 | 21.34 | | 17 | 50.01 | | 18 | 17.92 | | 19 | 24.47 | | 20 | 76.77 | | 21 | 20.96 | | 22 | 73.94 | | 23 | 34.38 | | 24 | 48.64 | | 25 | 42.43 | | 26 | 179.20 | | 27 | 15.94 | | 28 | 79.82 | | 29 | 19.32 |
|
| CDCl3 | | 01-Ha | | | 02-Ha | ~ 5.01 | | 03-He | 5.31 (ddd ) | | 04-Ha | | | 04-He | | | 05-H | | | 07-H | 5.85 (d ) | | 09-Ha | | | 11-Ha | | | 11-He | | | 12-Ha | | | 12-He | | | 15-Ha | | | 15-Hb | | | 16-Ha | | | 16-Hb | | | 17-H | | | 18-Me | 1.02 (s ) | | 19-Me | 0.85 (s ) | | 21-Me | 1.25 (s ) | | 22-H | ~ 4.98 | | 23-Ha | | | 24-H | | | 25-H | | | 27-Me | 1.28 (d ) | | 28-H | 4.10 (dq ) | | 29-Me | 1.41 (d ) |
| C5D5N | | 01-Ha | | | 02-Ha | 4.16 (m, w1/2=21.0) | | 03-He | 4.22 (m, w1/2=8.0) | | 04-Ha | | | 04-He | | | 05-H | 3.01 (dd, 13.2, 3.7) | | 07-H | 6.31 (d , 3.3) | | 09-Ha | 3.59 (dq ) | | 11-Ha | | | 11-He | | | 12-Ha | 2.65 (ddd, 12.9, 12.9, 5.1) | | 12-He | | | 15-Ha | | | 15-Hb | | | 16-Ha | | | 16-Hb | | | 17-H | 2.86 (brt, 8.8) | | 18-Me | 1.24 (s ) | | 19-Me | 1.07 (s ) | | 21-Me | 1.57 (s ) | | 22-H | 3.94 (m, w1/2=13.0) | | 23-Ha | | | 24-H | | | 25-H | 2.37 (dq, 11.1, 6.9) | | 27-Me | 1.36 (d, 6.9 ) | | 28-H | | | 29-H | 1.31 (d, 6.0) | | 29-Me | 1.31 (d, 6.0) |
|
| |
| M.P. | 164-166 °C (semihydrate) | | [α]D20 | +64.5° (c 1.0, H2O) | | IR (KBr) ν max (cm-1) | 3425, 1750 (gama-lactone), 1650 | | UV (EtOH) λ max (log ε) | 243 (4.11) |
| |
| TLC | NP-TLC silica, Rf 0.54 (CHCl3-EtOH-Me2CO 60:20:10 v/v/v), Rf 0.33 (CHCl3-MeOH 80: 20); RP-TLC on paraffin coated silica gel Rf 0.59 (MeOH-H2O 50:50); RP-TLC on C18-bonded silica gel Rf 0.44 (MeOH-H2O 65:35) | | GLC | Ret 19.5 min on 1.5% OV101 (0.9 m x 4.5 mm i.d.) at 285°C after silylation at 140°C for 60 h.(E : 5.85 min) | | HPLC | RP-HPLC Spherisorb-ODS ret 13.0 min, ACN-H2O 15:85, flow-rate 1 ml.min-1) (20E : 6.8 min); Nucleosil-ODS ret 9.5 min, ACN-H2O 15:85, flow-rate 1 ml.min-1) (20E : 4.4 min) | | LC | silica (preparative) CHCl3-MeOH (9:1) |
| |
Drosophila melanogaster BII cell assay: EC50 = 5.3 x 10-8M | Drosophila melanogaster BII cell assay: EC50 = 1.2 x 10-8M | Drosophila melanogaster imaginal disc assay: EC50 = 5.5 x 10-8M | Drosophila melanogaster Kc-H cell assay: EC50 = 2.8 x 10-8M | Galleria mellonella in vivo assay: ED50 = 3.8 ug/g | Sarcophaga bullata in vivo assay: ED50 = 10.4 ug/g | Dermestes vulpinus in vivo assay: ED50 = 21.0 ug/g | Musca assay: 100% (20-hydroxyecdysone = 100%) | Sarcophaga assay: 100% (20-hydroxyecdysone = 100%) |
| |
| First isolation | HIKINO, H. et al. (1968) Tetrahedron 24, 4895-4906 |
 | | General | IMAI, S. et al. (1969) Chem. Pharm. Bull. 17, 340-342 |
 | | General | TAKEMOTO, T. et al. (1971) Tetrahedron Lett., 3191-3194 |
 | | General | IKAN, R. et al. (1971) Phytochemistry 10, 1659-1661 |
 | | Bioactivities | CHIHARA, C.J. et al. (1972) J. Insect Physiol. 18, 1115-1123 |
 | | Bioactivities | CHERBAS, L. et al. (1980) W. Roux Arch. Devel. Biol. 189, 1-15 |
 | | Bioactivities | BERGAMASCO, R. et al. (1980) In: Progress in Ecdysone Research (Ed. J.A. Hoffmann), Elsevier/North Holland Biomed. Press, Amsterdam 299-324 |
 | | General | WILSON, I.D. et al. (1982) J. Chromatogr. 236, 224-229 |
 | | Bioactivities | SLÁMA, K. et al. (1993) Insect Biochem. Molec. Biol. 23, 181-185 |
 | | General | CALGANO, M.P. et al. (1995) Eur. J. Entomol. 92, 287-294 |
 | | Bioactivities | DINAN, L. et al. (1999) J. Computer-aided Mol. Design 13, 185-207 |
 | | Bioactivities | DINAN, L. et al. (2003) J. Insect Sci. 3:6 (pp. 11) |
 |
| |
Permanent link to this datasheet: CYASTERONE
|
|




![Wikipedia: Ajuga nipponensis [Lamiaceae (alt. Labiatae)]](/images/wikipedia.png)

