|
|
|
|
Year of first isolation: |
1970 |
| Formula: | C21H30O5 |
| Molecular weight: | 362 |
| Occurence in plants: |
Cyathula capitata [Amaranthaceae] »  ![Wikipedia: Cyathula capitata [Amaranthaceae]](/images/wikipedia.png)
Silene otites [Caryophyllaceae] »  ![Wikipedia: Silene otites [Caryophyllaceae]](/images/wikipedia.png)
Lychnis flos-cuculi [Caryophyllaceae] »  ![Wikipedia: Lychnis flos-cuculi [Caryophyllaceae]](/images/wikipedia.png)
Serratula tinctoria [Asteraceae] »  ![Wikipedia: Serratula tinctoria [Asteraceae]](/images/wikipedia.png)
Leuzea carthamoides [Asteraceae] »  ![Wikipedia: Leuzea carthamoides [Asteraceae]](/images/wikipedia.png)
|
| Occurence in animals: |
Calliphora stygia [Diptera] »  ![Wikipedia: Calliphora stygia [Diptera]](/images/wikipedia.png)
|
|
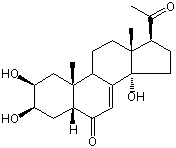
| |
| Canonical SMILES | CC(=O)C1CCC2(C1(CCC3C2=CC(=O)C4C3(CC(C(C4)O)O)C)C)O | Isomeric SMILES
[ PubChem: search | XML ]
[ ChemSpider: search ] | C1[C@]2([C@@H](C[C@H]([C@H]1O)O)C(=O)C=C1[C@@H]2CC[C@]2([C@]1(CC[C@@H]2C(=O)C)O)C)C » 
| | IUPAC Name | (2S,3R,5R,9R,10R,13R,14S,17S)-17-acetyl-2,3,14-trihydroxy-10,13-dimethyl-2,3,4,5,9,11,12,15,16,17-decahydro-1H-cyclopenta[a]phenanthren-6-one | | CAS-RN | 10162-99-9 | | PubChem CID | 441835 | InChiKey
[ ChemIDPlus: search ] | VNLQNGYIXVTQRR-NQPIQAHSSA-N | | InChI | InChI=1S/C21H30O5/c1-11(22)12-5-7-21(26)14-8-16(23)15-9-17(24)18(25)10-19(15,2)13(14)4-6-20(12,21)3/h8,12-13,15,17-18,24-26H,4-7,9-10H2,1-3H3/t12-,13+,15+,17-,18+,19-,20-,21-/m1/s1 |
| |
| CI-MS (NH3) m/z | 380 (M+H+NH3)+, 363 (M+H)+, 345, 327 | | EI-MS m/z (relative intensity %) | 362 (M)+, 344, 326, 301, 283 | | HR-MS | |
|
|
| C5D5N | | 01 | 37.8 | | 02 | 67.9 | | 03 | 67.9 | | 04 | 32.2 | | 05 | 51.2 | | 06 | 203.1 | | 07 | 122.0 | | 08 | 164.3 | | 09 | 34.4 | | 10 | 38.6 | | 11 | 21.0 * | | 12 | 31.8 ** | | 13 | 48.1 | | 14 | 83.9 | | 15 | 30.4 ** | | 16 | 21.9 * | | 17 | 59.4 | | 18 | 17.1 | | 19 | 24.3 | | 20 | 209.0 | | 21 | 31.2 |
|
| C5D5N | | 01-Ha | | | 01-Hb | | | 02-Ha | 4.09 (m ) | | 03-He | 4.15 (m ) | | 04-Ha | | | 04-He | | | 05-H | 2.90 (dd, 12, 4) | | 07-H | 6.09 (d , 2) | | 09-Ha | 3.46 (m ) | | 11-Ha | | | 11-He | | | 12-Ha | | | 12-He | | | 15-Ha | | | 15-Hb | | | 16-Ha | | | 16-Hb | | | 17-H | 3.52 (dd , 8, 8) | | 18-Me | 0.68 (s ) | | 19-Me | 1.00 (s ) | | 21-Me | 2.12 (s ) |
| CDCl3 | | 01-Ha | 1.41 (dd, ~13,10) | | 01-Hb | 1.86 | | 02-Ha | 3.89 (br m , w1/2=22) | | 03-He | 4.04 (br m, w1/2=8) | | 04-Ha | 1.63 | | 04-He | 1.84 | | 05-H | 2.44 (dd, 14, 4.5) | | 07-H | 5.84 (d, 2.5) | | 09-Ha | 3.02 (ddd, 11, 7, 2.5) | | 11-Ha | 1.64 | | 11-He | 1.86 | | 12-Ha | 2.08 | | 12-He | 1.55 | | 15-Ha | 2.0 | | 15-Hb | 1.6 | | 16-Ha | 2.27 | | 16-Hb | 1.84 | | 17-H | 3.27 (t, 8.5) | | 18-Me | 0.62 (s ) | | 19-Me | 0.97 (s ) | | 21-Me | 2.14 (s ) |
|
| |
| M.P. | 240-242°C (softening at 237°C) | | [α]D20 | | | IR (KBr) ν max (cm-1) | 3425 (OH), 1710 (acetyl), 1645 (cyclohexenone) | | UV (EtOH) λ max (log ε) | |
| |
| TLC | NP-TLC on silica gel : Rf 0.32 (CHCl3-EtOH 4:1); Rf 0.27 (EtOAc-MeOH-NH4OH 85:10:5); Rf 0.42 (CH2Cl2-EtOH 85:15). RP-TLC on paraffin-coated silica gel Rf 0.45 (MeOH-H2O 50:50); RP-TLC on C18-bonded silica gel Rf 0.42 (MeOH-H2O 65:35). | | GLC | Rt on 1.5% OV101 (0.9 m x 4 mm i.d.) at 285°C of 1.3 min after silylation at 140°C for 60 h. | | NP-HPLC | (Zorbax-Sil) Ret 15.2 min (isooctane-iPrOH-H2O 100:30:2); | | RP-HPLC | (Spherisorb-ODS2) Ret 9.7 min (solvent ACN-0.1% TFA in H2O, 23:77) (20-hydroxyecdysone 5.5 min). |
| |
Drosophila melanogaster BII cell assay: EC50 = 2.0 x 10-5M | Calliphora assay: 0% (20-hydroxyecdysone = 100%) |
| |
| General | SIDDALL, J.B. et al. (1967) J. Chem. Soc., Chem. Commun. 899- |
 | | General | GALBRAITH, M.N. et al. (1969) J. Chem. Soc., Chem. Commun. 1134-1135 |
 | | First isolation | HIKINO, H. et al. (1970) Steroids 16, 393-399 |
 | | General | HIKINO, H. et al. (1975) Chem. Pharm. Bull. 23, 125- |
 | | General | HIKINO, H. et al. (1975) Hoppe-Seyler´s Z. Physiol. Chem. 309-314 |
 | | Bioactivities | BERGAMASCO, R. et al. (1980) In: Progress in Ecdysone Research (Ed. J.A. Hoffmann), Elsevier/North Holland Biomed. Press, Amsterdam 299-324 |
 | | General | GIRAULT, J.-P. et al. (1990) J. Nat. Prod. 53, 279-293 |
 | | General | PETERSEN, Q.R. et al. (1993) Aust. J. Chem. 46, 1961-1964 |
 | | Identification | VOKÁČ, K. et al. (2002) Collect. Czech. Chem. Commun. 67, 124-139 |
 | | Bioactivities | DINAN, L. et al. (2003) J. Insect Sci. 3:6 (pp. 11) |
 |
| |
Permanent link to this datasheet: POSTSTERONE
|
|




![Wikipedia: Cyathula capitata [Amaranthaceae]](/images/wikipedia.png)

