|
|
|
|
Year of first isolation: |
1968 |
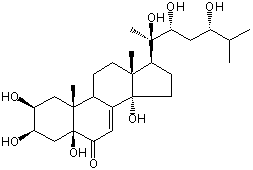
| Formula: | C27H44O8 |
| Molecular weight: | 496 |
| Occurence in plants: |
Podocarpus nakaii [Podocarpaceae] »  ![Wikipedia: Podocarpus nakaii [Podocarpaceae]](/images/wikipedia.png)
Dacrydium intermedium [Podocarpaceae] »  ![Wikipedia: Dacrydium intermedium [Podocarpaceae]](/images/wikipedia.png)
|
| Occurence in animals: |
|
|
|
| |
| Canonical SMILES | CC(C)C(CC(C(C)(C1CCC2(C1(CCC3C2=CC(=O)C4(C3(CC(C(C4)O)O)C)O)C)O)O)O)O | Isomeric SMILES
[ PubChem: search | XML ]
[ ChemSpider: search ] | C1[C@]2([C@@](C[C@H]([C@H]1O)O)(C(=O)C=C1[C@@H]2CC[C@]2([C@]1(CC[C@@H]2[C@](C)([C@@H](C[C@@H](C(C)C)O)O)O)O)C)O)C » 
| | IUPAC Name | (2S,3R,5S,9R,10R,13R,14S,17S)-2,3,5,14-tetrahydroxy-10,13-dimethyl-17-[(2R,3R,5S)-2,3,5-trihydroxy-6-methylheptan-2-yl]-1,2,3,4,9,11,12,15,16,17-decahydrocyclopenta[a]phenanthren-6-one | | CAS-RN | 26191-89-9 | | PubChem CID | 15249992 | InChiKey
[ ChemIDPlus: search ] | PXCOYCUFJMMDCP-CEPQAZHYSA-N | | InChI | InChI=1S/C27H44O8/c1-14(2)17(28)11-21(31)25(5,33)20-7-9-26(34)16-10-22(32)27(35)13-19(30)18(29)12-24(27,4)15(16)6-8-23(20,26)3/h10,14-15,17-21,28-31,33-35H,6-9,11-13H2,1-5H3/t15-,17-,18-,19+,20-,21+,23+,24+,25+,26+,27+/m0/s1 |
| |
| CI-MS (NH3) m/z | | | EI-MS m/z (relative intensity %) | (of the 20,22-benzylidene acetal) 584 (3), 379 (23), 362 (17), 206 (15), 143 (64), 135 (14), 134 (100), 125 (23), 107 (34), 105 (42), 91 (53) | | HR-MS | |
|
|
| C5D5N | | 01 | 34.6 | | 02 | 67.8 | | 03 | 69.7 | | 04 | 35.9 | | 05 | 79.7 | | 06 | 200.8 | | 07 | 119.8 | | 08 | 166.8 | | 09 | 38.2 | | 10 | 44.6 | | 11 | 21.3 | | 12 | 31.5 | | 13 | 48.0 | | 14 | 83.9 | | 15 | 32.0 | | 16 | 22.0 | | 17 | 40.8 | | 18 | 17.8 | | 19 | 17.0 | | 20 | 75.8 | | 21 | 21.5 | | 22 | 76.7 * | | 23 | 35.6 | | 24 | 77.5 * | | 25 | 34.0 | | 26 | 19.5 | | 27 | 17.0 |
|
| C5D5N | | 01-Ha | | | 01-Hb | | | 02-Ha | 4.12 | | 03-He | 4.03 | | 04-Ha | | | 04-He | | | 07-H | 6.17 (d, 2.5) 6.26 | | 09-Ha | | | 11-Ha | | | 11-He | | | 12-Ha | | | 12-He | | | 14-H | | | 15-Ha | | | 15-Hb | | | 16-Ha | | | 16-Hb | | | 17-H | | | 18-Me | 1.17 1.19 | | 19-Me | 1.12 1.13 | | 21-Me | 1.54 1.57 | | 22-H | | | 23-Ha | | | 23-Hb | | | 24-H | | | 25-H | | | 26-Me | 1.00 (d, 6) 1.02 (d, 6) | | 27-Me | 1.00 (d, 6) 1.02 (d, 6) |
|
| |
| M.P. | 270-272 °C | | [α]D20 | | | IR (KBr) ν max (cm-1) | 3375 (OH), 1668 (cyclohexenone), 1626 | | UV (EtOH) λ max (log ε) | 244 (4.04), 326 (2.0) |
| |
| HPTLC | | | TLC | Rf 0.23 (CHCl3-EtOH 4:1); Rf 0.38 (CHCl3-EtOH 5:1); Rf 0.65 (CHCl3-EtOH-Me2CO 6:2:1); Rf 0.59 (CHCl3-MeOH-Me2CO 6:2:1); Rf 0.05 (CH2Cl2-EtOH-Me2CO 16:1:4); Rf 0.45 (CH2Cl2-MeOH-C6H6 25:5:3). RP-TLC on paraffin coated silica gel Rf 0.34 (MeOH-H2O 50:50); RP-TLC on C18-bonded silica gel Rf 0.33 (MeOH-H22O 65:35). | | GLC | Ret 9.0 min (E 6.9 min) on 1.5% OV101 (0.9 m x 4.5 mm i.d.) at 285°C, following silyla-tion at 120°C for 5 h. | | HPLC | |
| |
Drosophila melanogaster Kc-H cell assay: EC50 = 2.8 x 10-9M | Calliphora assay: ~100% (20-hydroxyecdysone = 100%) |
| |
| First isolation | NAKANISHI, K. et al. (1968) Tetrahedron Lett. (9), 1105-1110 |
 | | General | KOREEDA, M. et al. (1970) Chem. Commun., 351-352 |
 | | General | HORN, D.H.S. et al. (1971) In: Naturally Occuring Insecticides (eds. Jacobson M. and Crosby D.G) Marcel Dekker, New York, pp. 333-459 |
 | | General | RUSSEL, G.B. et al. (1972) Aust. J. Chem. 25, 1935-1941 |
 | | General | BLUNT, J.W. et al. (1979) Aust. J. Chem. 32, 779-782 |
 | | Bioactivities | CHERBAS, L. et al. (1980) W. Roux Arch. Devel. Biol. 189, 1-15 |
 | | Bioactivities | BERGAMASCO, R. et al. (1980) In: Progress in Ecdysone Research (Ed. J.A. Hoffmann), Elsevier/North Holland Biomed. Press, Amsterdam 299-324 |
 | | General | WILSON, I.D. et al. (1985) J. Chromatogr. 318, 373-377 |
 | | General | BIELBY, C.R. et al. (1986) J. Chromatogr. 351, 57-64 |
 |
| |
Permanent link to this datasheet: PONASTERONE C
|
|




![Wikipedia: Podocarpus nakaii [Podocarpaceae]](/images/wikipedia.png)

