|
|
|
|
Year of first isolation: |
1995 |
| Formula: | C35H50O9 |
| Molecular weight: | 614 |
| Occurence in plants: |
Paxillus atrotomentosus [Fungi] »  ![Wikipedia: Paxillus atrotomentosus [Fungi]](/images/wikipedia.png)
|
| Occurence in animals: |
|
|
|
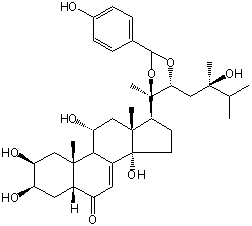
| |
| Canonical SMILES | CC(C)C(C)(CC1C(OC(O1)C2=CC=C(C=C2)O)(C)C3CCC4(C3(CC(C5C4=CC(=O)C6C5(CC(C(C6)O)O)C)O)C)O)O | Isomeric SMILES
[ PubChem: search | XML ]
[ ChemSpider: search ] | C1[C@]2([C@@H](C[C@H]([C@H]1O)O)C(=O)C=C1C2[C@@H](C[C@]2([C@]1(CC[C@@H]2[C@@]1([C@@H](C[C@@](C(C)C)(C)O)OC(O1)c1ccc(cc1)O)C)O)C)O)C » 
| | IUPAC Name | (2S,3R,5R,9R,10R,11R,13R,14S,17S)-2,3,11,14-tetrahydroxy-17-[(4R,5R)-5-(2-hydroxy-2,3-dimethylbutyl)-2-(4-hydroxyphenyl)-4-methyl-1,3-dioxolan-4-yl]-10,13-dimethyl-2,3,4,5,9,11,12,15,16,17-decahydro-1H-cyclopenta[a]phenanthren-6-one | | CAS-RN | | | PubChem CID | 15839568 | InChiKey
[ ChemIDPlus: search ] | CCUHDLIIVDDRTB-LAPRSRFCSA-N | | InChI | InChI=1S/C35H50O9/c1-18(2)33(5,41)17-28-34(6,44-30(43-28)19-7-9-20(36)10-8-19)27-11-12-35(42)22-14-23(37)21-13-24(38)25(39)15-31(21,3)29(22)26(40)16-32(27,35)4/h7-10,14,18,21,24-30,36,38-42H,11-13,15-17H2,1-6H3/t21-,24+,25-,26+,27-,28+,29+,30?,31-,32+,33?,34+,35+/m0/s1 |
| |
| EI-MS m/z (relative intensity %) | (M)+ (0), 456 (1), 438 (3), 423 (2), 420 (1), 413 (3), 404 (2), 395 (3), 354 (7), 342 (10), 337 (6), 326 (14), 299 (21), 281 (10), 157 (26), 139 (19), 121 (96), 107 (30), 87 (70), 69 (68), 55 (68), 43 (100) | | FAB-MS m/z (relative intensity %) | 637 (M+Na)+ (3), 615 (M+H)+ (2), 597 (2), 475 (7), 457 (5), 439 (3), 422 (8), 400 (20), 391 (4), 383 (2), 355 (4), 345 (2), 327 (3), 312 (6), 299 (4), 284 (4), 263 (7), 250 (7), 188 (17), 121 (39), 109 (61), 92 (36), 83 (42), 69 (72), 55 (100) | | HR-MS | (M+H)+, found 615.7342, calc. 615.7439 |
|
|
| CD3OD | | 01 | 39.09 | | 02 | 68.96 | | 03 | 68.58 | | 04 | 33.31 | | 05 | 52.79 | | 06 | 206.71 | | 07 | 122.89 | | 08 | 165.32 | | 09 | 42.92 | | 10 | 39.88 | | 11 | 69.44 | | 12 | 43.43 | | 13 | a | | 14 | 85.04 | | 15 | 31.84 | | 16 | 22.67 | | 17 | 50.72 | | 18 | 18.54 | | 19 | 24.62 | | 20 | 85.73 | | 21 | 23.52 | | 22 | 81.30 | | 23 | 39.38 | | 24 | 75.23 | | 25 | 39.55 | | 26 | 18.17 | | 27 | 17.52 | | 28 | 22.13 | | C6H4-OH | 131.22, 129.45 (2), 115.86 (2), 159.39 | | O-CH-O | 105.26 |
|
| CD3OD | | 01-Ha | 1.38 (dd, 13.0, 12.0) | | 01-He | 2.59 (dd, 13.0, 4.2) | | 02-Ha | 4.01 (ddd, 12.0, 4.2, 3.3) | | 03-He | 3.95 (q ) | | 04-Ha | 1.79 (dt, 14.2, 13.2, 2.5) | | 04-He | 1.69 (dt, 14.2, 4.0, 3.5) | | 05-H | 2.34 (dd, 13.2, 4.0) | | 07-H | 5.82 (dd, 2.7, 0.8) | | 09-H | 3.15 (dd, 9.0, 2.7) | | 11-He | 4.10 (ddd,10.8, 9.0, 6.0) | | 12-Ha | 2.22 (dd, 12.2, 10.8) | | 12-He | 2.14 (dd, 12.2, 6.0) | | 15-Ha | ~ 2.05 | | 15-Hb | 1.63 (m ) | | 16-Ha | ~ 2.05 | | 16-Hb | ~ 2.05 | | 17-H | 2.42 (dd, 9.0, 8.5) | | 18-Me | 0.871 (s ) | | 19-Me | 1.051 (s ) | | 21-Me | 1.280 (s ) | | 22-H | 4.18 (d, 9.4) | | 23-Ha | 1.80 (dd, 14.8, 9.4) | | 23-Hb | 1.56 (bd, 14.8) | | 25-H | 1.74 (h, 6.8) | | 26-Me | 0.968 (d, 6.8) | | 27-Me | 0.934 (d, 6.8) | | 28-Me | 1.135 (s ) | | C6H4-OH | 6.77 (m ), 7.29 (m ) | | O-CH-O | 5.73 (s ) |
|
| |
| M.P. | | | [α]D20 | | | IR (KBr) ν max (cm-1) | 3409 (OH), 1657 (cyclohexenone), 1617, 1518 (C=C), 1102 (C-O | | UV (EtOH) λ max (log ε) | |
| |
| NP-HPLC | column Silasorb 600 (4 x 250 mm), flow-rate 0.8 ml.min-1, solvent Et2O-CH3CN-H2O (780:190: 30), Ret 20.3 min (20E 31.7 min) | | HPTLC | | | TLC | | | GLC | | | RP-HPLC | column Separon SIX C18 (4 x 250 mm), solvent MeOH-H2O, linear gradient 10 to 70 % MeOH in 50 min, flow-rate 0.6 ml.min-1, Ret 57.2 min (20E 42.7 min). |
| |
Drosophila melanogaster BII cell assay: EC50 = 3.0 x 10-7M |
| |
| |
Permanent link to this datasheet: PAXILLOSTERONE 20,22-p-HYDROXYBENZYLIDENE ACETAL
|
|




![Wikipedia: Paxillus atrotomentosus [Fungi]](/images/wikipedia.png)

